Understanding the shelf life of bakery products
The baked snack industry is a constantly growing market valued at $440 billion. These products typically cover a range of textures, colors, and flavors, and it is critical that they remain safe while maintaining their expected texture and sensory properties. One of the most important factors influencing the quality and shelf life of bakery products is water activity. Water activity control can help prevent or minimize various degradative events such as rancidity, microbiological spoilage, staling, or changes in texture due to water migration.
Theory of water activity
Water activity is defined as the Energy status of water in a system and is rooted in the fundamental laws of thermodynamics through Gibb’s freeenergy equation. It represents the relative chemical potential energy of water as dictated by the surface, colligative, and capillary interactions in a matrix. Practically, it is measured as the partial vapor pressure of water in a headspace that is at equilibrium with the sample, divided by the saturated vapor pressure of water at the same temperature. Water activity is often referred to as the ‘free water’, but since ‘free’ is not scientifically defined and is
interpreted differently depending on the context, this is incorrect. Free water gives the connotation of a quantitative measurement, while water activity is a qualitative measurement of the relative chemical potential energy. Rather than
a water activity of 0.50 indicating 50% free water, it more correctly indicates that the water in the product has 50% of the energy that pure water would have in the same situation. The lower the water activity then, the less the water
in the system behaves like pure water. For baked snacks, water activity is measured by equilibrating the liquid phase water in the sample with the vapor phase water in the headspace of a closed chamber and measuring the Equilibrium Relative Humidity (ERH) in the headspace using a sensor. The relative humidity can be determined using a resistive electrolytic sensor, a chilled mirror sensor, or a capacitive hygroscopic polymer sensor. Instruments from Novasina, like the Labmaster NEO, utilize an electrolytic sensor to determine the ERH inside a sealed chamber containing the sample. Changes in ERH are tracked by changes in the electrical resistance of the electrolyte sensor. The advantage of this approach is that it is very stable and resistant to inaccurate readings due to contamination, a particular weakness of the chilled mirror sensor. The resistive electrolytic sensor can achieve the highest level of accuracy and precision with no maintenance and infrequent calibration. Sampling for water activity testing can be particularly challenging for bakery products because they tend to be too large to fit in the sample cup and are often multi-component While water activity is an intensive property that provides the energy of the water in a system, moisture content is an extensive property that determines the amount of moisture in a product. Water activity and moisture content, while related, are not the same measurement. Moisture content is typically
determined through loss-on-drying as the difference in weight between a wet and dried sample. For baked snacks, moisture content provides a standard of identity and an expected mouthfeel but does not determine if the product is microbially safe. Water activity and moisture content are related through the moisture sorption isotherm. Table 1 shows that different baked snacks can have similar water activities, but very different moisture contents. Obviously, the moisture content associated with a safe water activity will be different for each product and as will be demonstrated in the next section, should never be
relied on as an indicator of microbial safety.
WATER ACTIVITY AND TEXTURE/CRISPINESS
Bakery products are known for their pleasing texture and taste and each product has its own unique organoleptic requirements. The most common mode of failure in baked products is an unexpected change in texture or flavor. For example, a butter biscuit should be crispy, and a cake should show a good mixture of firmness and moisture. Figure 1 shows the water activity range for baked snacks and the expected texture for each region. Changes in water activity will result in changes in texture and each baked product has an optimal water activity range where the texture and taste will be ideal. The key then to prolonging the shelf life of these products is to manufacture them to that ideal water activity and maintain that water activity during storage and transport with effective packaging.

Figure 1. Examples of baked snacks covering the range of water activity and the expected texture profile for each snack
Many bakery products contain multiple components such as a cream filling or an icing covering. For These products, moisture migration between components can lead to undesirable texture changes or even susceptibility to microbial growth. Moisture moves from high water activity to low water activity, so the way to prevent moisture migration and its accompanying consequences is to match the water activity of the components. The icing, cake, and cream filling of a snack cake all need to be manufactured to the same water activity before being combined and then, even if their moisture content is different, there will be no moisture migration between the components

Figure 2. Diagram showing that to avoid moisture migration, the water activity of multi-component snack cakes need to match, regardless of their moisture content.
WATER ACTIVITY AND CHEMICAL STABILITY
The largest category of baked snacks
contains products whose water activities
are less than 0.70 aw, so microbial
spoilage is not the most likely event
that will end shelf-life. For products
that need to be in the 0.50-0.70 aw,
range to maintain their desirable texture,
chemical degradation is a strong
candidate because reactions rates are
at a maximum. In general, as water
activity increases so do reaction rates,
but lipid oxidation is unique in that the
reaction rate also increases at very low
water activity (1). Examples of reactions
that can result in product degradation
and end of shelf life include staling,
Maillard browning (changes in color
and flavor), lipid oxidation (rancidity),
enzymatic (changes in texture, color,
and flavor), and vitamin break down
(nutritional quality loss). Since lowering
water activity to slow reactions is not an
option, as it would change the texture,
instead, other interventions are typically
warranted such as reformulation to
remove reactants, inclusion of oxygen
absorbers, or modified atmosphere
packaging.
Deep fried products such as potato
chips pose a unique problem. The water
activity of these products is quite low
at less than 0.10 aw. The assumption
may be that they should have extremely
long shelf life since their water activity
is so low. However, as mentioned
earlier, the rate of lipid oxidation that
leads to rancidity uniquely increase at
very low water activity (Figure 3). Since
these products contain high levels of
lipids in the form of fat, they are very
susceptible to oxidation. Consequently,
these products are typically packed in
a nitrogen flush to remove oxygen and
have a limited shelf life.
When changes in flavor or odor due to
chemical reactions is what is ending
shelf life, the time required for the reaction
to have progressed to the point
of unacceptability at a given water
activity and temperature will be the
product’s shelf life. If the rate constants
for these reactions at several different
storage conditions are determined, a
predictive model can be used to estima-
te the time needed for the reaction to
proceed to an unacceptable level under
any storage conditions. To do this, the
progress of the reaction will need to be
tracked using some type of quantitative
assessment. Examples of methods for
quantifying common reactions include:
Lipid Oxidation/Rancidity
- Peroxide values
- Tbar values
- Oxygen consumption
- Sensory
Browning Reactions - Color changes
- Sensory
- Formation of reaction products
Vitamin Loss - Test kits (ELISA)
- HPLC
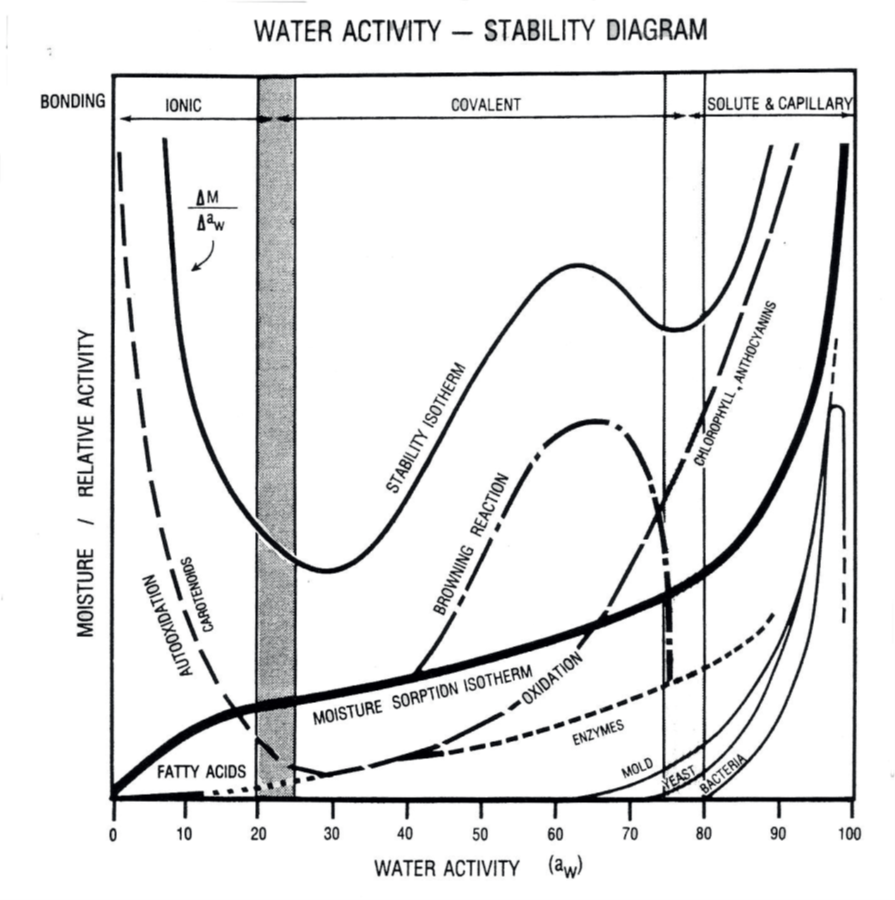
While there are examples of shelf life models in the literature, the only fundamental model that includes both water activity and temperature is hygrothermal time (2). It is derived from a form of the Eyring (3) equation for rate change and Gibbs equation for free energy and is given by where T is the temperature (K), R is the gas constant (J mol-1 K-1), Ea is the activation energy (J mol-1), B is the molecular volume ratio, aw, is the water activity, and r0 is the rate at the standard state. In practice, the values for B, Ea/R and r0 will be unique to each situation and are derived empirically through least squares iteration. Once the constants are known, any temperature and water activity can be used with the hygrothermal time model to determine rate of oxidation at those conditions and hence the shelf life that the product will remain acceptable to the consumer.
Water Activity and Microbial Growth *****
The actual organism that will proliferate at a given water activity varies with each organism having a lower water activity limit below which it will not grow. A list of the water activity lower limits for growth for common spoilage organisms can be found in Table 2 (4). These growth limits indicate that all pathogenic bacteria stop growing at water activities less than 0.87 while the growth of common spoilage
yeasts and molds stops at 0.70 aw, which is known as the practical limit. Only xerophilic and osmophylic organisms can grow below 0.70 aw, and all microbial growth stops at water activities less than 0.60. Other intrinsic factors such as pH impact microbial growth as well. Baked snacks in this range are not considered unsafe because the growth of molds and yeasts does not cause foodborne illness.
However,the growth of non-pathogenic organisms does typically render the product undesirable to a consumer and is considered to have ended theshelf life of the product. Consequently, the water activity must be reduced to below 0.70E, or other interventions are needed to prevent mold growth.
| Microorganism | aw limit | Microorganism | aw limit |
| Clostridium botulinum E | 0.97 | Penicillum expansum | 0.83 |
| Pseudomonas fluorescens | 0.97 | Penicillum islandicum | 0.83 |
| Escherichia coli | 0.95 | Debarymoces hansenii | 0.83 |
| Clostridium perfringens | 0.95 | Aspergillus fumigatus | 0.82 |
| Salmonella spp. | 0.95 | Penicillum cyclopium | 0.81 |
| Clostridium botulinum A B | 0.94 | Saccharomyces bailii | 0.8 |
| Vibrio parahaemoliticus | 0.94 | Penicillum martensii | 0.79 |
| Bacillus cereus | 0.93 | Aspergillus niger | 0.77 |
| Rhizopus nigricans | 0.93 | Aspergillus ochraceous | 0.77 |
| Listeria monocytogenes | 0.92 | Aspergillus restrictus | 0.75 |
| Bacillus subtilis | 0.91 | Aspergillus candidus | 0.75 |
| Staphylococcus aureus (anaerobic) | 0.9 | Eurotium chevalieri | 0.71 |
| Saccharomyces cerevisiae | 0.9 | Eurotium amstelodami | 0.7 |
| Candida | 0.88 | Zygosaccharomyces rouxii | 0.62 |
| Staphylococcus aureus (aerobic) | 0.86 | Monascus bisporus | 0.61 |
Table 2. Water activity lower limits for growth for common spoilage organisms.
For chocolate products, water activity is measured by equilibrating the liquid phase water in the sample with the vapor phase water in the headspace of a closed chamber and measuring the Equilibrium Relative Humidity (ERH) in the headspace using a sensor. The equilibrium relative humidity (water activity) can be determined using a resistive electrolytic sensor, a chilled mirror sensor, a tunable diode laser, or a capacitive hygroscopic polymer sensor. Instruments from Novasina, like the Labmaster NEO, utilize a resistive electrolytic sensor to determine the ERH. Changes in ERH are tracked by changes in the electrical resistance of the electrolyte sensor.
Chocolate confections typically do not contain volatiles – except in certain liquor-based formulations – so volatile filters are generally not required during water activity testing. However, a key challenge in measuring the water activity of chocolate products is the extended test time. Due to their high fat content, chocolate confections require longer equilibration periods, as the movement of high-energy water molecules into the vapor phase occurs more slowly. Taking readings before true equilibrium is reached, can result in inaccurate measurements, potentially compromising product safety and quality.
Equilibrium in water activity instrumentation is usually determined by the rate of change in water activity reaching a predefined threshold. The stricter this threshold, the greater the confidence in capturing the true water activity value, but this also increases the test duration. Some instruments attempt to shorten test times by using a less stringent default end-of-test setting, which may not be adjustable by the user. Novasina prioritizes the integrity of water activity testing and therefore allows users to set their preferred level of stringency for the end-of-test criteria. Users also have the option to follow the ISO 18787 standard method for water activity testing, ensuring consistency and reliability in their measurements (https://www.iso.org/standard/63379.html).
To demonstrate the potential differences resulting from various end-of-test (stability) settings, Novasina scientists conducted water activity tests on a range of products using both the less stringent Fast Stability setting and the more stringent Slow Stability setting on the Novasina LabMaster NEO (see Table 1). The Fast setting is designed to deliver quicker results by requiring that consecutive water activity readings differ by no more than 0.001 aw for 2 minutes. In contrast, the Slow setting ensures a more robust equilibrium by requiring the same level of stability over a 6-minute period.

While the Fast setting can significantly reduce test time, it may come at the expense of accuracy. If the results obtained using the Fast setting differ from those using the Slow setting by less than 0.01 aw, it is generally acceptable to use the Fast setting to expedite testing. However, for critical applications where precision is paramount, the Slow setting provides greater confidence in capturing the true water activity value.
Using a less stringent end-of-test setting should never be done without first evaluating the potential difference in results, something only possible if the instrument allows users to adjust these settings. Novasina recommends that when beginning water activity testing for a new product, users should first run the test using the Fast Stability setting. Then, without opening the lid, leave the sample in the instrument for up to an hour. During this time, the LabMaster NEO continues to update the water activity reading in real time, while also displaying the value obtained using the Fast setting. This allows users to assess whether the difference is significant enough to warrant switching to a more stringent stability setting.
As shown in Table 1, some chocolate products such as chocolate syrup exhibited minimal differences in average water activity (less than 0.01 aw) between the Fast and Slow settings, despite the longer test time required by the Slow setting. However, other products, like chocolate hazelnut spread, showed a much larger difference of 0.09 aw. These results demonstrate how a QA lab can determine which products are suitable for faster testing and which require longer equilibration times to ensure accurate measurements.
Water Activity and Microbial Growth
Microbial safety is a primary concern for all food products. Proper processing is essential to reduce microbial load and prevent the proliferation of harmful microorganisms. Water activity plays a critical role in controlling microbial growth, as it directly affects an organism’s ability to reproduce. When a microorganism encounters an environment with lower water activity than its internal level, it experiences osmotic stress – losing water to the environment as it moves toward a lower energy state (2). This water loss reduces turgor pressure and slows down metabolic activity.
To continue reproducing, the microorganism must lower its internal water activity below that of its surroundings, allowing water to re-enter the cell. It attempts this by concentrating solutes internally. The effectiveness of this strategy varies by organism, meaning each has a unique minimum water activity threshold below which it cannot grow (2,3). Table 2 lists the lower water activity limits for growth of common spoilage organisms.
According to Table 2, most chocolate confections listed in Table 1 have water activity levels below the minimum required for microbial growth. Therefore, microbial spoilage is unlikely to be the primary factor limiting shelf life. However, it is important to note that some pathogenic bacteria – most notably Salmonella – can survive for extended periods in low water activity environments like chocolate confections by forming spores (4). As a result, low water activity should never be considered a kill or sterilization step. If chocolate is later used as an ingredient in a product with a higher water activity matrix, dormant Salmonella cells may resume growth and pose a health risk.

Microbial Concerns for Chocolate Syrups
While the hard chocolate confections listed in Table 1 have water activity levels below the growth limits for microorganisms, the chocolate syrup products exhibit significantly higher water activity. Both syrups exceed the growth threshold for molds and could support mold growth if left exposed to airborne spores. Notably, the zero-sugar syrup has a much higher water activity than the syrup containing sugar. The sugar-based syrup has a water activity of 0.827 aw, which is below the growth limits for all pathogenic bacteria and is considered shelf stable. In contrast, the zero-sugar syrup has a water activity of 0.937 aw, exceeding the growth limits for both Staphylococcus and Listeria. According to the FDA Food Code, a product with this level of water activity can only be considered shelf stable if its pH is below 5.6 (see Figure 1).
This increase in water activity highlights the challenges faced by formulators when developing healthier alternatives. While reducing sugar content offers health benefits, it can also compromise product safety. Sugar acts as a natural humectant, controlling water activity by providing multiple water-binding sites that lower the energy of water in the product. Replacing sugar with alternative sweeteners, each with different chemical properties, reduces the number of water-binding sites. Unless total moisture is also reduced, which is often impractical in products like syrups due to viscosity requirements, water activity will increase. If this change in water activity is not properly documented and adjustments are not made to maintain product safety, the reformulated product could pose a health risk. Therefore, water activity must always be monitored during formulation, especially when removing polar ingredients such as sugar or salt.
Water Activity and Chocolate Flavor

Flavor is one of the most important quality attributes of chocolate confections. Consumers expect a flavor profile that is consistent with the product type, and any deviation can make the product undesirable, effectively ending its shelf life. The naturally low water activity of most chocolate confections contributes to their long shelf life by slowing down chemical reactions that could alter flavor. However, oxidation of fats remains a key concern, as it can lead to the development of off-flavors associated with rancidity.
Water activity influences the rate of lipid oxidation in chocolate products. Unlike most food reactions, lipid oxidation increases at both low and high water activity levels, as illustrated in Figure 2. This means there is an optimal water activity range, known as the monolayer value, where the rate of oxidation is minimized. For most chocolate products, this value is around 0.35 aw, which aligns with the water activity levels of many products listed in Table 1. To ensure flavor stability and extend shelf life, chocolate products should be processed to achieve this ideal water activity range. Incorporating water activity testing into the quality assurance protocols of chocolate manufacturers is essential for maintaining product integrity and consumer satisfaction.
Water Activity and Chocolate Bloom
In addition to flavor, texture and appearance are critical quality attributes for chocolate confections. Consumers expect an appealing visual presentation and, even more importantly, a specific mouthfeel. Dark chocolate is typically associated with a crisp snap and firmer texture, while milk chocolate is expected to deliver a smooth, creamy mouthfeel.
The most common degradative reaction affecting both appearance and texture is chocolate bloom, which can occur as either sugar bloom or fat bloom. Water activity is most closely linked to sugar bloom, which happens when the surface water activity of chocolate increases due to exposure to high humidity and then returns to its original level. At elevated water activity, sugars dissolve into solution and later recrystallize on the surface as the water activity drops. This results in a white, dusty coating of sugar crystals, giving the chocolate a rough texture and negatively impacting its visual appeal. It also alters the melting behavior during consumption, interfering with the expected creamy mouthfeel.
To prevent sugar bloom, chocolate products must be processed to their ideal water activity, as determined through water activity testing. Additionally, it is essential to maintain consistent storage conditions to prevent fluctuations in water activity that could trigger bloom formation.



